Last week, Galmed Pharmaceuticals (NASDAQ:GLMD) shares climbed as high as $9.90 per share this week, up approximately 7% on the day after the company released updated phase IIb results for Aramchol, the company’s treatment for NASH (non-alcoholic steatohepatitis).
Galmed reported the 600mg arm of Aramchol achieved NASH resolution without a worsening of fibrosis, and a fibrosis stage reduction without worsening of NASH. Both endpoints are considered to be required for regulatory approval by the FDA in phase III trials.
Galmed was one of the inaugural reports included for SanaCurrents members that took us up on our exclusive limited time offer through GeoInvesting.

The other company for which we offered a report, Eidos Therapeutics (NASDAQ:EIDX), also experienced an increase in share prices this week leading up to and after reporting positive clinical data.
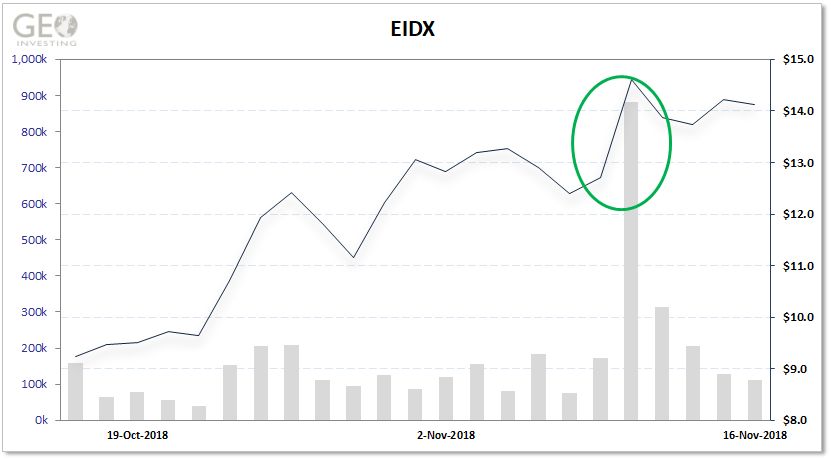
SanaCurrents assigned a superior sentiment rating, its highest, to Eidos and the stock rose 17% on the day after the results were announced. Galmed received an advantageous rating, the third highest by SanaCurrents, and the stock climbed 7%. By assigning these sentiment ratings, we believe it gives a feel for the degree of how positively the market may react to the upcoming news.
What do we think these results mean for the future? EIDX and GLMD shares remain attractive companies for us to track over the next weeks and months because there are no strong therapies on the market for the diseases they treat, ATTR cardiomyopathy and NASH. As a SanaCurrents subscriber, you will get clinical updates that we believe will allow you to make informed decisions before the next EIDX or GLMD catalysts are announced.
As our service progresses into the next phase, we plan to inform SanaCurrents premium members with more reports assigning sentiment ratings to binary events that have historically affected stock performance in biopharmaceutical and medical device companies.
Based on feedback we’ve already received, we’re shaping the service to include more options for current and future members, like ongoing model portfolios to enable them to see how taking a measured approach can mitigate risk.
In addition, you will receive many more companies to track similar to Eidos and Galmed. The advance notice of catalysts, along with the independent research from SanaCurrents, is intended to help you keep tabs on the anticipated growth in biopharmaceuticals and medical devices, two sectors that are projected to expand in this highly competitive market.
I look forward to sharing more of our research with you. Below were the two reports sent to premium SanaCurrents members.
Check out the current SanaCurrents offering here and begin to receive our probability sentiments on select biotech and medical device stocks.
~Bill Langbein